Polymeric Film

HAIRCARE

DECORATIVE COSMETICS

SKINCARE

SUNCARE
HAIRCARE
Polymers designed for hair care products that provide long-lasting shine, strong hold and essential protection for healthy hair.
DECORATIVE COSMETICS
Film-forming solutions for makeup and nail polish products, delivering long-lasting adhesion, non-transfer properties and flawless coverage.
SKINCARE
Invisible films formulated for skincare that offer hydration, protection, and skin barrier enhancement for a smoother, healthier appearance.
SUNCARE
Advanced dispersions that improve sunscreen effectiveness, offering superior UV protection while ensuring long-lasting skin defense.
ECOPOL TECH FILM FORMING POLYMERS
Mastering Polymeric Film
Our film-forming polymers shape the future of Beauty
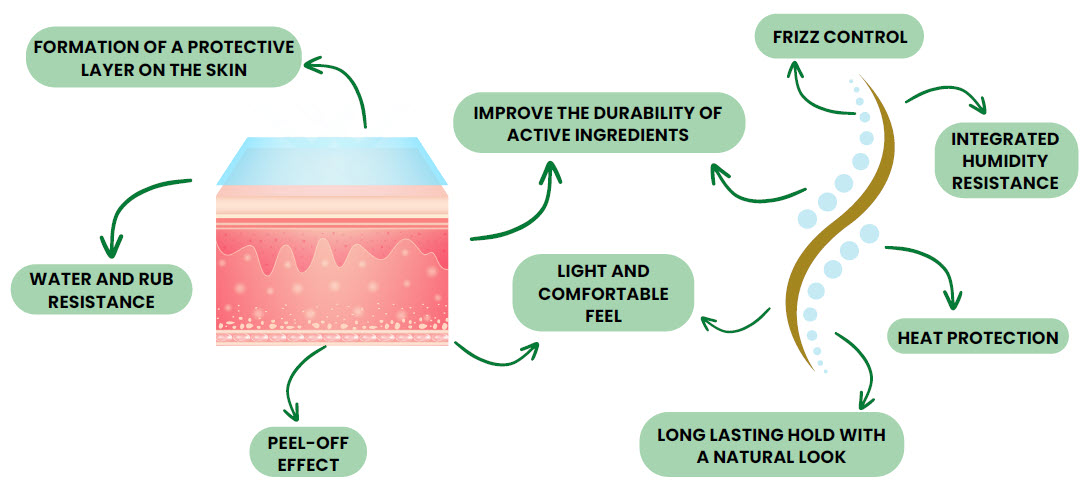
Skin Protective layer
Formation of a protective layer on the skin. This layer offers strong water and rub resistance. Additionally, the compound enables a peel-off effect.
Hair Protective layer
Formation of a protective layer on the hair. Long lasting and performant protection and hair control, with integrated humidity resistance and heat control.
Hair Conditioning
A protective polymer layer on the hair offers significant frizz control. As hair-conditioning our polymers deliver long lasting hold with a natural look.
Future proof care-tech
We deliver solutions for Personal Care, both skin-care and hair-care products feel light and comfortable while improving the durability of active ingredients





















